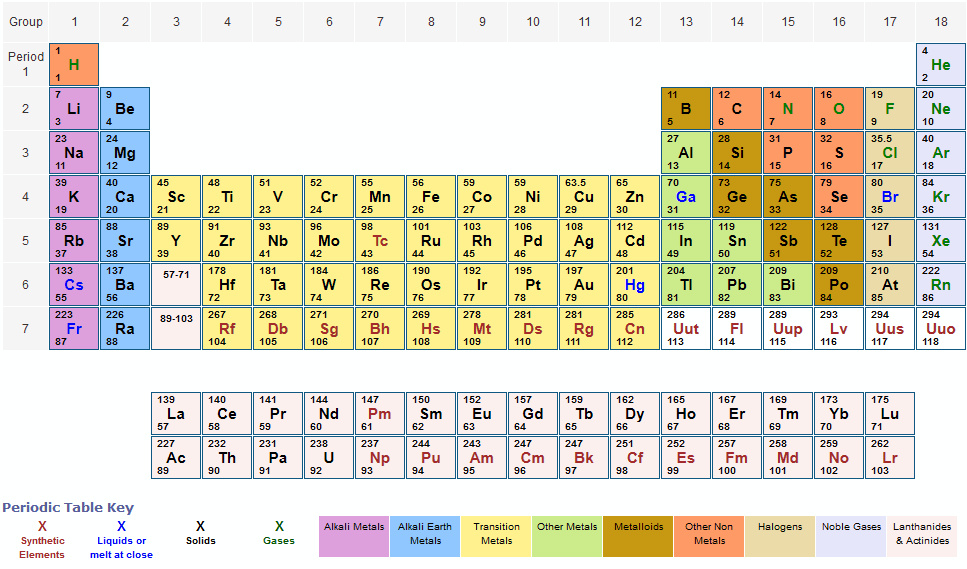
We have excluded elements above 108 on this periodic table for the simple reason that the larger elements don’t really exist in the real world. You might find a table of elements showing element 115 or even 118 elements total. There is a Netflix documentary about this topic entitled Bob Lazar: Area 51 & Flying Saucers. The elements above 92 are strange, like for example element 115 that was reportedly found at Area 51. The parenthesis are a scientific way of saying this number is guesstimated, at best, and we really don’t know much about these elements because they are artificial and tend to disintegrate rapidly. You might note in the periodic table the mass numbers are in parenthesis for elements 93 and above. At least you won’t be finding any in this lifetime. 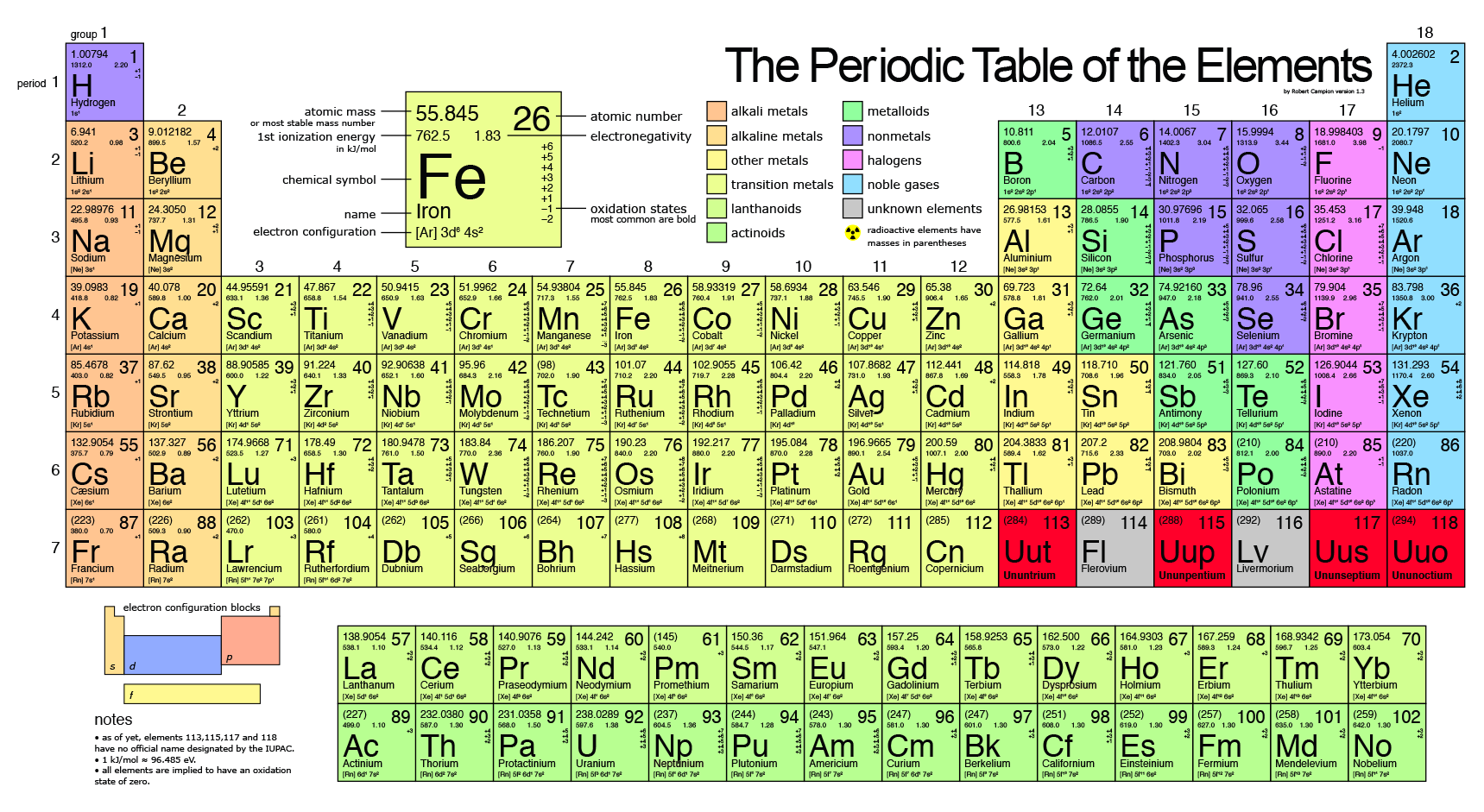
As such, elements above 92 don’t really exist, except under special laboratory conditions. All elements above atomic number 92 are created artificially in laboratories, and generally they are extremely unstable and tend to disintegrate rapidly. The molar mass of a substance, also often called molecular mass or molecular weight (although the definitions are not strictly identical, but it is only sensitive in very defined areas), is the weight of a defined amount of molecules of the substance (a mole) and is expressed in g/mol. As such, it’s highly unstable and unable to exist for very long. 
It is element 94, making it bigger than uranium. In 1940, a new element, plutonium, was created in a laboratory. Element 92 is uranium, the biggest element that occurs naturally on Earth. Before the year 1940, it was believed that only 92 elements existed. (See the original work of Dmitri Mendeleev here.) At that time, scientists were still discovering new elements almost every year. The original table of the elements, credited to Dmitri Mendeleev in 1871, had only 56 elements. Thus, the formula mass of calcium hydrogen carbonate is 117.10 amu and the molar mass of calcium hydrogen carbonate is 117.10 grams per mole (g/mol).The table of elements shown here has 108 elements. However when talking about a mole of an ionic compound we will still use the term molar mass. This is because there are no individual molecules in ionic compounds. For compounds that are not molecular (ionic compounds), it is improper to use the term “molecular mass” and “ formula mass” is generally substituted. The molar mass of the N 2 molecule is therefore 28.02 g/mol. This is referred to as the molecular mass and the molecular mass of any molecule is simply the sum of the atomic masses of all of the elements in that molecule. For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) = 28.02 amu. For a molecule (for example, nitrogen, N 2) the mass of molecule is the sum of the atomic masses of the two nitrogen atoms. The concept of molar mass can also be applied to compounds. Further, if you have 16.00 grams of oxygen atoms, you know from the definition of a mole that your sample contains 6.022 x 10 23 oxygen atoms. proton mass fine-structure constant 1/137.035 999 Rydberg constant 10 973 731.569 m1 Boltzmann constant 1.380 65 x 1023 J K1 c h e molar gas constant 8.314 5 J mol1 K1 electron volt 1.602 177 x 10-19 J (exact) 0.510 998 950 MeV 13.605 693 eV R R c R R hc mec 2 mp 1.602 176 634 x 1019 C 1.672 622 x 1027 (exact)kg 3.289 841. For example, the atomic mass of an oxygen atom is 16.00 amu that means the molar mass of an oxygen atom is 16.00 g/mol. Generalizing this definition, the molar mass of any substance in grams per mole is numerically equal to the mass of that substance expressed in atomic mass units. By definition, the molar mass of carbon-12 is numerically the same, and is therefore exactly 12 grams. We have defined a mole based on the isotopic atomic mass of carbon-12. 
The atomic mass of an element is the relative average of all of the naturally occurring isotopes of that element and atomic mass is the number that appears in the periodic table. ) a carbon atom with six proton and six neutrons in its’ nucleus, surrounded by six electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
